
If ammonium ions are present, the ions will convert to ammonia gas. We start the process by adding dilute sodium hydroxide to our substance, we then go on to gently heat this combination. You can find ammonium ions in a variety of different substances such as ammonium solution or any ammonium salt like ammonium chloride. The final test that we are going to explore is how to detect ammonium ions (NH4+). Table 2: Different halide ions and the coloured precipitate they produce when reacting with nitric acid and silver nitrate. If a yellow precipitate is produced, iodide ions are present, if a cream precipitate is produced, bromide ions are present, and if a white precipitate is produced, chloride ions are present. To distinguish between the different halides, we will observe the colour of the precipitate produced. 
This test also allows us to determine exactly which halide ion is present, so there is no need to carry out any additional tests. We then add some silver nitrate and if a precipitate forms, this means halide ions are present. This removes any carbonate ions produce, which can affect our results. Our next test is to detect the different halide ions, these are chloride ions, Cl - bromide ions, Br - and iodide ions, I -.įor this reaction, we start by adding some dilute nitric acid to the substance we are testing. Lithium ions, Li +, produce a crimson flame colour, sodium ions (Na +) produce a yellow flame colour, potassium ions, K +, produce a lilac flame colour, calcium ions (Ca 2+) produce an orange-red flame colour, and copper ions, Cu 2+, produces a green flame colour. Now to explore the different colours you will see and match them to the right metal ion. If a metal compound has a mixture of metals, some of the flame colours may be dominated by others. You will see a specific colour, and you can compare this to when we explore the different colours.Now hold the loop in the blue flame of a Bunsen burner.Then dip it into the acid again and then into the metal compound that is being tested.First, get a nichrome wire loop and dip it into concentrated hydrochloric acid, then heat it.During this test, each metal cation produces a different coloured.īefore we explore those different colours let us go through how we experiment: The flame test is quite exciting and you might even carry it out in your classroom. Metal ions are quite interesting because they are different bright colours, this makes it easier to distinguish between them and also makes it more fun to learn. To detect metal ions we can use two types of tests using a flame and using sodium hydroxide. This means that they are positively charged and have lost electrons. Finally, we will go through the test for ammonium ions.Then we will explore the test for halide ions.We will then go on to the test for sulphate ions.After, we will explore the test for carbonate ions.We will first go over the test for metal ions this will include the flame test and using sodium hydroxide solution.We will be going through this test, as well as many others. 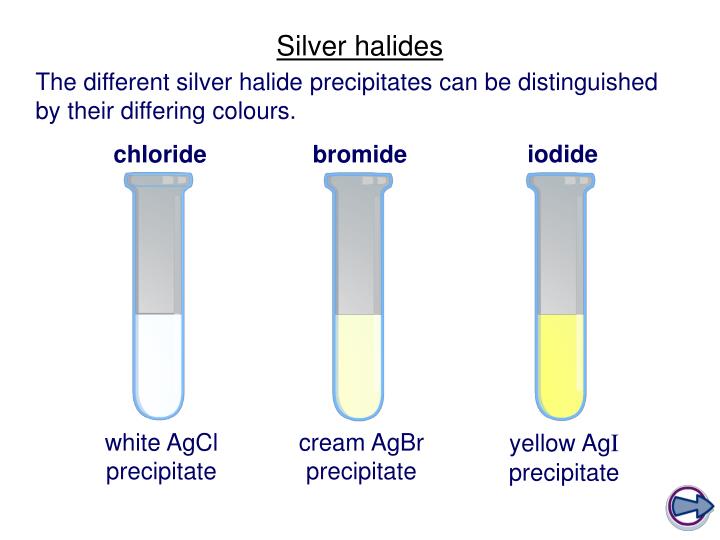
When looking at swimming pools, for example, chlorine is added to kill any bacteria, but how can we ensure that it is in there? This is where testing for ions comes in, as we can take a sample of water and test for the chloride ions. However, there are some chemicals that we can ingest but this is to a certain point. This implies that bromine is not a better oxidizing agent than chlorine or, in other words, that chlorine is a better oxidizing agent than bromine.The water that you drink, the pool that you swim and the food that you eat all have many different chemicals. There will be no color change the hexane layer will remain orange.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
